Acid cam qt1/6/2023  
Vonoprazan (Takecab) was released to the market in Japan in February 2015. It was found to have satisfactory effects and a good safety profile in clinical studies of gastric and duodenal ulcers, reflux esophagitis, NSAID-associated ulcers, and H. With these efforts, vonoprazan (TAK-438), a potassium-competitive acid blocker (P-CAB), was developed. To overcome the aforementioned unmet needs, alternative formulations of conventional PPIs and new H +, K +-ATPase inhibitors have been established. 12– 14 Finally, although it requires an acidic environment for activation, PPIs are unstable in acidic conditions, 15 so enteric coating is needed. 10, 11 Third, its effects at night are not satisfactory. 9 Second, the effects of PPIs are influenced by cytochrome P450 (CYP) 2C19 polymorphism. 5– 7 Reflux symptoms of GERD are not sufficiently relieved after the first dose of PPIs in two-thirds of patients because of its slow onset of the action, 8, 9 and one-half of patients still have symptoms even after 3 days of treatment. 4 First, it takes several days to show maximal effect. 1– 3 Although PPIs have been used for more than a quarter-century as a first-line treatment for these diseases, it has become clear that there are some issues in need of improvement ( Table 1). Conventional PPIs with a benzimidazole structure irreversibly inhibit hydrogen potassium (H +, K +)-ATPases, which produce acid in gastric parietal cells and more strongly block acid secretion compared to histamine H2 receptor antagonists. Proton pump inhibitors (PPIs) have often been used for acid-related diseases including gastroesophageal reflux disease (GERD), gastric and duodenal ulcers, non-steroidal anti-inflammatory drug (NSAID)-associated ulcers, and Helicobacter pylori eradication therapy. Keywords: Anti-inflammatory agents, non-steroidal, Esophagitis, H +, K +-exchanging ATPase, Helicobacter pylori, Potassium-competitive acid blocker Thus, vonoprazan may address the unmet medical need for the treatment of acid-related diseases. It does not require an acidic environment for activation, has long-term stability at the site of action, and has satisfactory safety and tolerability. Vonoprazan accumulates in parietal cells under both acidic and neutral conditions. 
Vonoprazan does not require enteric coating as it is acid-stable, and it can be taken without food because it is quickly absorbed. It is currently indicated for the treatment of gastric and duodenal ulcers, reflux esophagitis, and prevention of low-dose aspirin- or nonsteroidal anti-inflammatory drug-related gastric and duodenal ulcer recurrence in Japan. 
Vonoprazan was recently innovated as a novel, orally active P-CAB. Potassium-competitive acid blockers (P-CABs) were developed and have beneficial effects including rapid, long-lasting, and reversible inhibition of the gastric hydrogen potassium ATPase, the proton pump of the stomach. Alternative formulations of conventional PPIs have been developed to overcome these problems however, these drugs have only introduced small advantages for controlling acid secretion compared to conventional PPIs. However, they have a number of limitations including slow onset of action, influence by cytochrome P450 polymorphisms, unsatisfactory effects at night, and instability in acidic conditions. Conventional proton pump inhibitors (PPIs) are used as a first-line therapy to treat acid-related diseases worldwide. 
0 Comments

The vulnerability is triggered when the user opens a crafted MP3 file or loads a remote stream URL that is mishandled by Clementine. #Symantec encryption desktop 10.3.2 download trial code#It allows an authenticated (low privileged) attacker to execute remote code on the target server within the context of application's permissions (SYSTEM).Ĭlementine Music Player through 1.3.1 (when a GLib 2.0.0 DLL is used) is vulnerable to a Read Access Violation on Block Data Move, affecting the MP3 file parsing functionality at memcpy+0x265. jsp filename for a file that begins with audio data bytes. Is it searching for a signature, at the low-level, or is it searching at the file-level? It looks like it's combing through sectors, so I'm thinking it's looking for a particular signature to attempt to piece together the PGP instrumentation of the disk.Multiple Cross-Site Request Forgery (CSRF) vulnerabilities in Simon Ward MP3 jPlayer plugin <= 2.7.3 at WordPress.Īn arbitrary file upload vulnerability exists in albumimages.jsp in Quicklert for Digium 10.0.0 (1043) via a. My question is where is the original MBR stored on the disk? Somewhere on the hidden track (read: track 0) or within one of the encrypted partitions? When you back up the MBR, I'm assuming it backs up the Master Partition Table and disk signature, in addition to the Master Boot Code (basically, all 512-bytes).Īlso, when running the PGP WDE -recover command, I see it searches throughout the disk. When the disk is instrumented, the original MBR is restored. ).īasically, Symantec uses up to the first 63 sectors (track 0) for their PGP MBR. I think I've finally got a decent technical understanding of how PGP interacts with the MBR, thanks to the link here (.  
but there is some important data on the drive so I haven't given up yet! Help! This started scanning each sector of the drive, but bombs out consistently at a particular sector with the below error. Great, I thought, now I'll try the -recover command line option. This is what I see (Disk 1 is the old HDD attached via USB): So I plugged the disk in to a machine running the same version (10.3.0) of PGP using a SATA-USB cable, and tried the command line. I tried the PGP recovery boot disk first, but this did not detect the drive. I've spent a couple of days scouring the forums and found this post which mirrors my problem exactly. Booting the system then gave the message "missing operating system". Not thinking, I ran fixmbr which obviously blew away the WDE boot loader. Running the diagnostic tools in there indicated that the Master Boot Record of the laptop was to blame. #Symantec encryption desktop 10.3.2 download trial password#A couple of days ago Windows stopped booting correctly, and immediately after entering my PGP password the machine booted into the Windows recovery environment. My company mandates the use of Symantec PGP Whole Disk Encryption, so my Windows 7 laptop is duly encrypted. #Symantec encryption desktop 10.3.2 download trial install#* Can I install it after the disk is encrypted? * What if I install PGP WDE on the Windows 2012? Will it make any difference? #Symantec encryption desktop 10.3.2 download trial drivers#* Is there any drivers that Windows 2012 needs to read the file system better? When the D-drive become encrypted, the Windows server 2012 VHD will be encrypted too. Unencrypted.Ĭurrently the C-drive is partly encrypted and Windows server 2012 can't read the filesystem anymore and lists the partition as RAW format. It may not be related to me fiddeling with the Windows 2012 evaluation VHD, but the correlation is strong.ġ laptop with a 500GB drive, split into 3 partitions:Ĭ: contains my provided Windows 7 and is currently 25% encrypted.ĭ: contains my Win2012.vhd file, which has been inserted in the Windows bootloader and can be selected on system start. 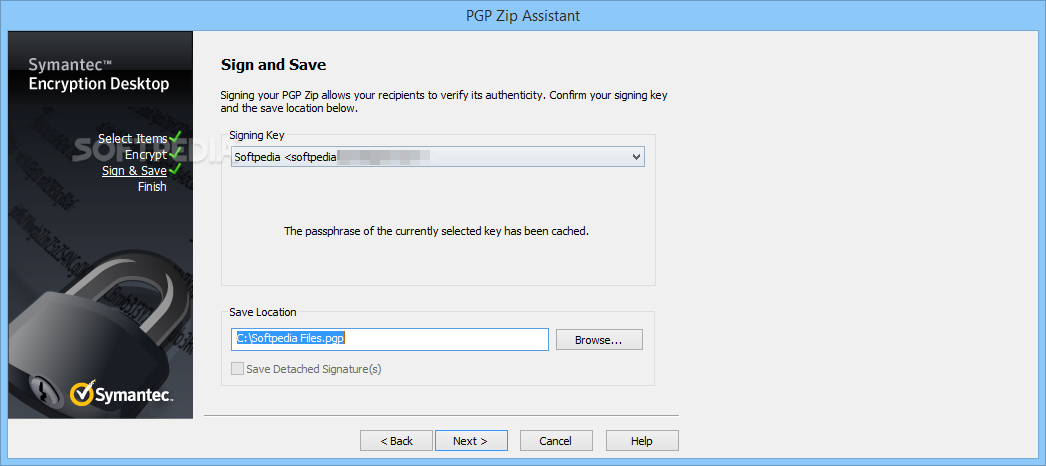
the reason I just got my computer reinstalled, is that the C-drive became too corrupted to even start. I just paused the encryption process as I forsee some issues with reading the disk, as I tried this on an already encrypted disk earlier and had some bad crashes and load errors. It comes with PGP WDE 10.2.0 MP3, which is progressing nicely and encrypting everything. I have my laptop, which has been provided for me at work.  Ethyl acetate molar mass1/6/2023  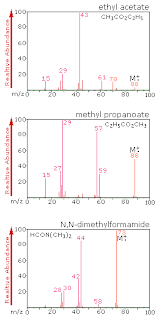
Acknowledgements This work was supported by the National Natural Science Foundation of China under Grant Nos. Ethyl Acetate has applications as a solvent in inks for flexographic and rotogravure printing. Therefore, few-cycle femtosecond laser mass spectra have strong potentials in analyzing molecular structure as well as the single photon ionization mass spectra. Ethyl Acetate is used in a variety of coating formulations such as epoxies, urethanes, cellulosics, acrylics and vinyls. 
Ethyl acetate is the most common ester in wine, being the product of the most common volatile acetic acid and the ethyl alcohol generated during the fermentation. Ethyl acetate is used primarily as a solvent and diluent, being favored because of its low cost, low toxicity, and agreeable odor. The results are compared with the corresponding dichloromethane mobile phases. It is also prepared in industry using the Tishchenko reaction, by combining two equivalents of acetaldehyde in the presence of an alkoxide catalyst. Identification of ETHYL ACETATE Chemical Compound 88.10512 g/mol ethyl acetate CCOC(C)O InChI1S/C4H8O2/c1-3-6-4(2)5/h3H2,1-2H3 XEKOWRVHYACXOJ-UHFFFAOYSA-. SummaryThe reversed phase behaviour of high molecular mass polystyrenes was investigated on C18 bonded phase columns with ethyl acetate-methanol and ethyl acetateacetonitrile mobile phases. Ethyl acetate is synthesized in industry mainly via the classic Fischer esterification reaction of ethanol and acetic acid. 
It is colorless liquid has a characteristic sweet smell and is used in glues, nail polish removers, decaffeinating tea and coffee, and cigarettes. Molar mass of CH 3 COOCH 2 CH 3, Ethyl Acetate is 88.10512 g/mol. Ethyl acetate is the ester of ethanol and acetic acid. Molar Mass Calculator measurement compact unit conversion calculator.  AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
